Ethically and legally sourced organs are always in demand. The high demand and low supply result in an unfortunately high mortality rate, not due to transplantation, but due to waiting for one. In India, the gap between demand and available organs is staggering—roughly 150,000–200,000 patients are waiting for kidney transplants, yet only a few thousand transplants occur.
Here comes the idea of xenotransplantation: where one transplants organs, tissues, or cells from one species into another. In recent years, this idea has shifted from science fiction [Frankenstein] to clinical reality. In 2025–2026 alone, xenotransplants have moved from an idea to the first structured clinical trials, thus offering a potential solution to one of medicine’s most persistent problems.
This article unveils what xenotransplantation actually is, how pig organs are genetically edited, the real-world anecdotes, the challenges that remain, and what the future might look like.
Join Weekly Health Newsletter
Every week, I will share with you information about Natural Health, Diet, Exercise, Yoga & Wellness.
DEFINING XENOTRANSPLANTATION
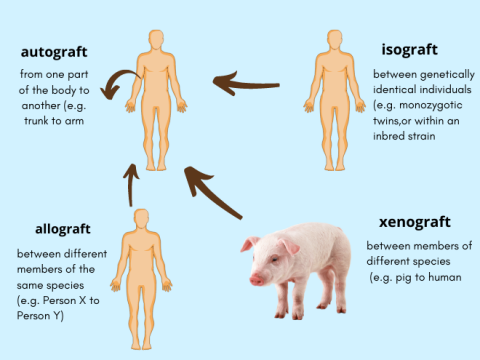
Types of Transplants or Grafts
Transplantation is the process of transferring cell, tissue or organ from one being to another. Now, they might be:
- Autograft: Tissue or organ is transplanted from one part of a person’s body to another part of the same person.
- Isograft: Tissue is transferred between genetically identical individuals (e.g., identical twins).
- Allograft: A tissue or organ is transferred from one person to another (same species, not genetically identical).
- Xenograft: Transplantation of organs or tissues between different species
For practical reasons, the primary animal used is the pig, though some research uses primates or other species. In modern medicine, xenotransplants are being developed for kidneys (for end-stage renal disease), hearts (for severe heart failure), livers (for acute or chronic liver failure), Islet cells (for type 1 diabetes), and skin and corneal tissues (for burns and eye‑disease patients).
BUT WHY PIGS?
Just like how Mendel selected Pisum sativum [Sweet pea] for his genetic experiments, pigs seem to have been selected for cultivating xenotransplants. Here are the reasons:
- Similar size and anatomy
- Pig hearts and kidneys are roughly the same.
- Their physiology is also close enough that early‑stage transplants can restore basic function of the organ
- Rapid and scalable breeding

- Thanks to their high reproductive capacity, a large number of pigs can be procured in a shorter amount of time.
- This means, in theory, that thousands of “organ-ready” pigs could be bred on demand, unlike the limited supply of human donor organs.
- Advanced genetic‑editing tools
- CRISPR and other gene‑editing technologies have made it possible to make tailor-made organs fitting an individual.
- In landmark cases, pig kidneys and hearts used in humans carried up to 69 different genetic edits.
Because of this combination—anatomy, scalability, and genetic programmability—pigs have become the workhorse species for modern xenotransplantation.
GENETICALLY ENGINEERED PIGS
In short, we can say there are 3 major goals for genetic engineering:
Prevention of Transplant Rejection
Human immune systems has the ability to detect “self” from “non-self” antigens. Pig antigens like α‑Gal (Galactose‑α‑1,3‑galactose) and other glycan antigens are flagged as “foreign”. This led to hyperacute graft rejections within minutes in early xenotransplant attempts.
The scientists now use CRISPR to overcome this problem. Recent successful pig‑kidney cases involved multiple antigen‑knockout edits plus human‑gene insertions to fine-tune compatibility.
But preventing rejection isn’t just about removing pig markers. The compatibility can also be increased by the addition of human genes.
- CD46, CD55, CD59: Regulate the complement system
- Thrombomodulin, EPCR (endothelial protein C receptor): Reduce abnormal clotting within the graft [major cause of early failure]
- CD47: Prevents phagocytosis of pig cells
- HO‑1 (heme‑oxygenase‑1): Has anti-inflammatory and protective effects on blood vessels inside the organ
In one landmark pig‑kidney transplant (2024–2025), the donor pig carried 69 different genetic edits—a mix of knockouts, knock-ins, and knockdowns. This genetically engineered organ functioned in a human recipient for over 270 days (nearly nine months).
Tackling Thrombosis
Thrombotic Microangiopathy
One of the biggest issues in early xenotransplantation was thrombotic microangiopathy. It is the development of multiple clots in small blood vessels of the organ, leading to ischaemia and death of the tissue.
The solution has been twofold:
- Engineering pig organs with human clot-regulating genes (thrombomodulin, EPCR, CD47, etc.).
- Providing immunosuppressive and anticoagulant drug regimens around the time of transplant.
Chances of Zoonotic Infections
Pigs are a host to several different viruses, including porcine endogenous retroviruses (PERVs). These viruses are integrated in the pig’s DNA and can, in theory, infect human cells, raising concerns about long-term safety.
Recent advances include:
- Gene‑editing PERVs out of the pig genome using CRISPR.
- Protocols to check for any viral activation or transmission.
So far, no PERV transmission has been definitively documented in human xenotransplant cases, but the risk remains a key focus of regulatory scrutiny.
REAL-WORLD XENOTRANSPLANT CASES
Now let’s look at the most recent cases of xenotransplants…
Pig Kidney
The first truly durable pig-kidney transplant in a human was a compassionate use case in 2024–2025. A patient with end-stage kidney disease and no hope of a human donor received a kidney from a 69‑gene-edited pig.
Key outcomes:
- Normal filtration function, reducing the need for dialysis.
- The patient reported improved energy, better appetite, and fewer complications tied to chronic dialysis.
- The graft survived for over 270 days, far longer than any previous pig‑kidney xenotransplant.
In 2026, the first FDA‑sanctioned clinical trial for pig‑to‑human kidney xenotransplantation began in the United States at major centers including NYU Langone, Massachusetts General Hospital, and Johns Hopkins.
These trials are small, closely monitored, and follow strict protocols for patient selection, immunosuppression, and follow‑up.
Organizations like the National Kidney Foundation have also highlighted that clinical trials for pig‑to‑human kidney transplantation are now officially underway.
Pig Heart
In the early 2020s, the first pig‑to‑human heart transplants under compassionate‑use protocols made headlines. Some recipients survived for weeks to months, far longer than was expected.
The associated risks were immense, but each case provided critical data:
- Response of human immune cells
- Better drug combinations to balance rejection prevention and side effects.
- Better protocols for selecting recipients
In 2025–2026, the International Society for Heart and Lung Transplantation (ISHLT) published a consensus statement that lays out strict guidelines for clinical cardiac xenotransplantation. These include:
- Which human antirejection drugs are acceptable?
- How to monitor for rejection and infection?
- How to manage informed consent and long-term follow-up?
This document is essentially a “blueprint” for bringing pig‑heart transplants out of the emergency room and into structured clinical practice.
Pig Liver
In 2025, a world‑first pig‑to‑human liver xenotransplant in a living patient was reported in China. The recipient, a patient with severe liver failure, received a 6‑gene‑edited miniature‑pig liver.
This liver functioned for several weeks, providing metabolic support and buying time for the patient’s condition to stabilize. While the graft did not last years, it demonstrated that pig livers can replace a human’s liver, even for a short duration.
Reports from the European Association for the Study of the Liver (EASL) and other liver‑disease societies now frame pig‑liver xenotransplants as a potential bridge for acute‑liver‑failure patients who would otherwise die within days.
Other Pig Transplants
Researchers are also exploring pig‑derived islet‑cell transplants for type 1 diabetes.
Scientists are also looking at:
- Pig‑skin grafts for burn patients (providing temporary coverage until human‑skin grafts can be prepared).
- Pig‑corneal transplants for patients with corneal scarring or infection.
RESPONSE OF HUMAN IMMUNE SYSTEM
Xenotransplantation is a constant battle against the immune system. The body’s reaction to a pig organ typically follows a sequence:
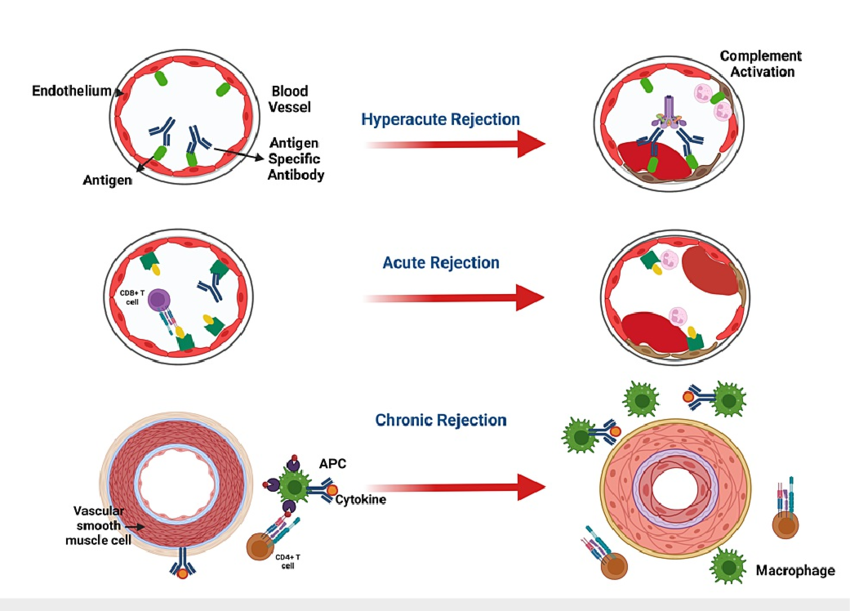
Types of Graft Rejection
Hyperacute and Acute Rejection
In early attempts, the immune system would destroy pig organs within minutes to hours via hyperacute rejection. This happened because:
- Human antibodies bound to pig‑specific antigens.
- The complement system activation, causing massive blood‑clotting and organ destruction.
Modern gene editing and immunosuppression have reduced the incidence dramatically, but low‑level immune activity often still occurs over days to months.
Chronic Immune Reactions
Even with genetic modifications, the human immune system continues to interact with the pig graft in the long run.
A 2026 immunology paper analyzing the first long‑lasting pig‑kidney xenograft showed that:
- Certain immune‑cell subsets remained active even months after transplant.
- Gene‑expression patterns in the graft revealed subtle changes reflective of ongoing immune stress.
Thrombosis and Coagulation
Thrombotic microangiopathy remains one of the toughest issues. Pig‑human blood‑clotting systems are not perfectly aligned, so:
- Microclots form in small vessels inside the graft.
- Over time, these clots block blood flow and cause organ failure.
Solutions now include:
- Genetic insertion of human-like clot-regulating genes (thrombomodulin, EPCR, CD47).
- Careful use of anticoagulant drugs around the transplant window.
These interventions have significantly reduced early clotting-related failures in recent xenotransplants.
Infection and Viral Risks
The final concern is infection, especially from pig-specific viruses. Regulators and clinicians are carefully monitoring:
- Porcine endogenous retroviruses (PERVs): As mentioned earlier, CRISPR is being used to delete or inactivate PERVs from donor pigs.
- Other pathogens: Pigs are raised in strict biosecure facilities, and organs are screened for bacteria, parasites, and common viruses.
Post‑transplant, patients undergo regular testing for any sign of viral or microbial transmission.
To date, there is no confirmed case of PERV transmission from pig‑to‑human xenografts, but the risk is treated as a top‑priority issue.
Porcine endogenous retroviruses
SIGNIFICANCE IN HUMAN PATIENTS
The primary driver of xenotransplantation is the uneven demand and supply chain. Xenotransplantation offers a possible way to close this gap by:
- Providing on‑demand organs not dependent on sudden accidents or deaths.
- Reducing waiting times and mortality for patients on dialysis, in heart‑failure clinics, or on liver‑support systems.
- Offering time for more options: a pig organ can keep someone alive and stable until a human organ becomes available.
For patients, the benefits are profound, including and not limited to improved quality of life, greater predictability, and potential longevity.
CHALLENGES AND ETHICAL QUESTIONS
Despite the promise, xenotransplantation is still high‑risk, experimental surgery with major open questions.
Long‑Term Survival and Function
Even the best‑case pig‑kidney xenografts have so far lasted months, not decades. Long‑term outcomes—like 10‑ or 20‑year survival—are still unknown.
Immunosuppression and Side Effects
Xenotransplant recipients need strong immunosuppressive drugs to suppress their immune systems enough to accept the pig organ. These drugs:
- Increase susceptibility to infections.
- Raise the risk of cancer.
- Can cause kidney damage, diabetes, and other metabolic problems.

Ethical Considerations
Xenotransplantation raises serious ethical issues:
- Is it acceptable to breed and modify pigs solely for human organ harvesting?
- Patients in end‑stage disease may feel pressured to accept experimental procedures with uncertain outcomes.
- Will xenotransplants become another tool for the wealthy, while the poor remain on dialysis or in heart‑failure clinics?
Cost and Scalability
Right now, xenotransplantation is extremely expensive: gene editing, pathogen‑controlled breeding, surgery, and long‑term monitoring all carry high costs.
Scaling this technology to hundreds of thousands of patients worldwide will require dramatic cost reductions and technological simplification.
REFERENCES
Tao P and Zhou K (2025) Recent progress in pig-to-human kidney xenotransplantation. Front. Immunol. 16:1735113. doi: 10.3389/fimmu.2025.1735113
Shirini, Kasra et al. “Xenotransplantation Literature Update: January-June 2025.” Xenotransplantation vol. 32,4 (2025): e70072. doi:10.1111/xen.70072
Clinical Trials for Pig-to-Human Kidney Transplantation Are Here. National Kidney Foundation. August 05, 2025.
Guilherme T. Ribas et al, Immune profiling in a living human recipient of a gene-edited pig kidney, Nature Medicine (2026). DOI: 10.1038/s41591-025-04053-3
U.S. FDA page summarizing regulatory concerns, infection risks, and oversight of animal‑to‑human transplants. Link.
International Society for Heart and Lung Transplantation document on rules for patient selection, immunosuppression, and monitoring in pig‑heart xenotransplants. 20 February 2026.
National Kidney Foundation‑style page explaining what xenotransplantation is, how it works, and where trials stand. Link.

0 comments