Pancreatic cancer is one of the most aggressive and lethal malignancies. The majority of cases are due to Pancreatic Ductal Adenocarcinoma [85–90% of all pancreatic cancers]. A new drug Daraxonrasib, developed by Revolution Medicines, has shown about a two-fold increase in survival benefit during its Phase 3 Clinical Trials.
This article deals with a short introduction of pancreatic cancer, its connection to RAS and the new developments to its treatment.
Join Weekly Health Newsletter
Every week, I will share with you information about Natural Health, Diet, Exercise, Yoga & Wellness.
INTRO TO PANCREATIC CANCER
Pancreatic cancer is still one of the most challenging diseases to tackle in medicine, and at its core lies pancreatic ductal adenocarcinoma (PDAC)—the most common and aggressive type. Despite advances in oncology, PDAC continues to carry a poor prognosis, majorly due to its silent progression and late diagnosis.
PDAC arises from the ductal epithelial cells of the pancreas and accounts for nearly 85–90% of all pancreatic cancers. It typically affects individuals between the ages of 60 and 80 and is among the leading causes of cancer-related deaths worldwide.
Risk Factors
Major Factors are :
- Smoking (most important modifiable risk factor)
- Chronic pancreatitis
- Diabetes mellitus (new-onset in elderly = red flag)
Genetic factors are :
- KRAS mutation (>90%)
- TP53, CDKN2A, SMAD4
- Syndromes:
- BRCA mutation
- Lynch syndrome
- Peutz-Jeghers syndrome
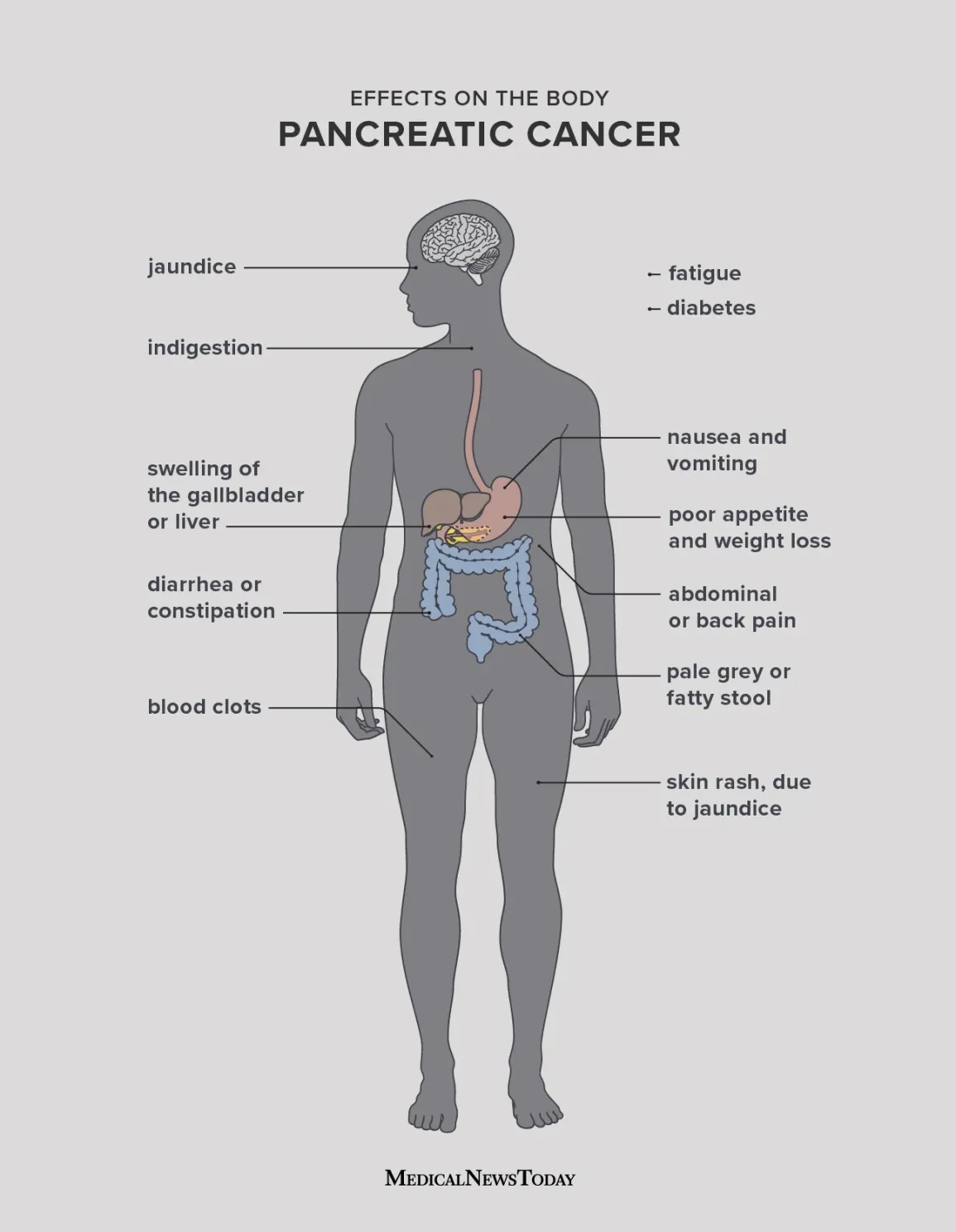
Pathogenesis
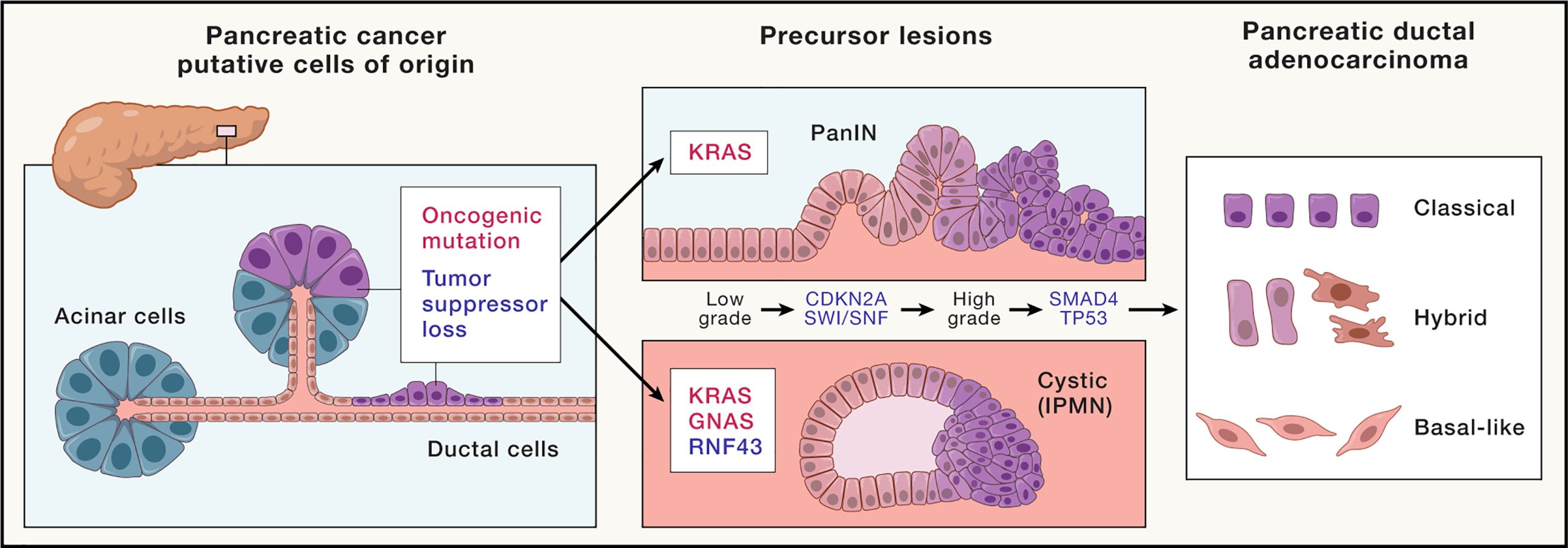
The disease often develops through precursor lesions known as PanIN (pancreatic intraepithelial neoplasia), progressing gradually to invasive cancer.
Clinical Features
Tumors in the head of the pancreas commonly present with:
- Painless jaundice
- Dark urine and pale stools
- Itching (pruritus)
Tumors in the body or tail may present later with:
- Epigastric pain radiating to the back
- Unintentional weight loss
- Loss of appetite
A classic clinical sign is Courvoisier’s sign—a palpable, non-tender gallbladder in a jaundiced patient.
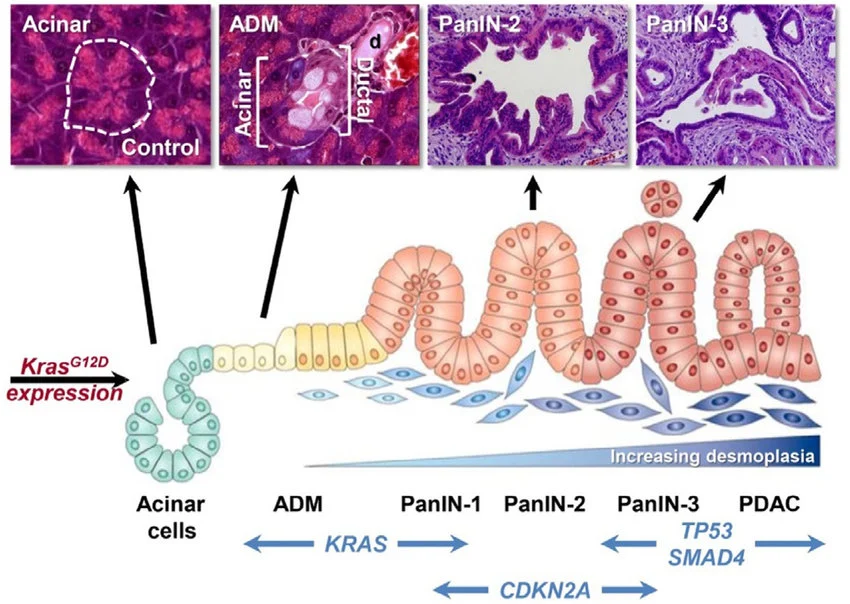
Diagnosis and Evaluation
Early detection is rare, but when suspected, the following are key:
- Contrast-enhanced CT (CECT): first-line imaging
- CA 19-9: tumor marker used for monitoring (not screening)
- Endoscopic ultrasound (EUS) with biopsy: confirms diagnosis
Staging focuses heavily on whether the tumor is resectable, which determines treatment strategy.
TNM Staging of Cancer
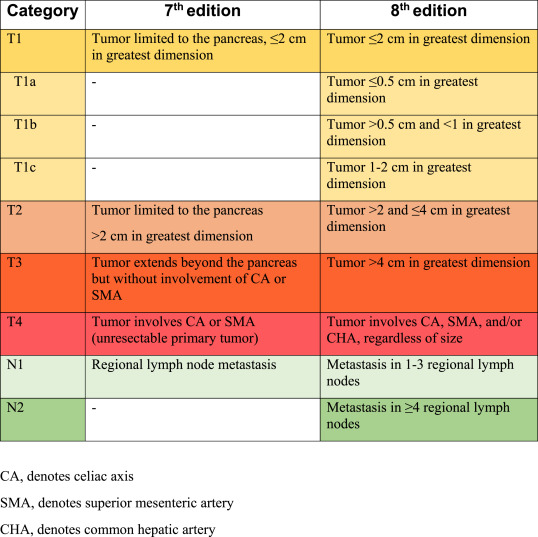
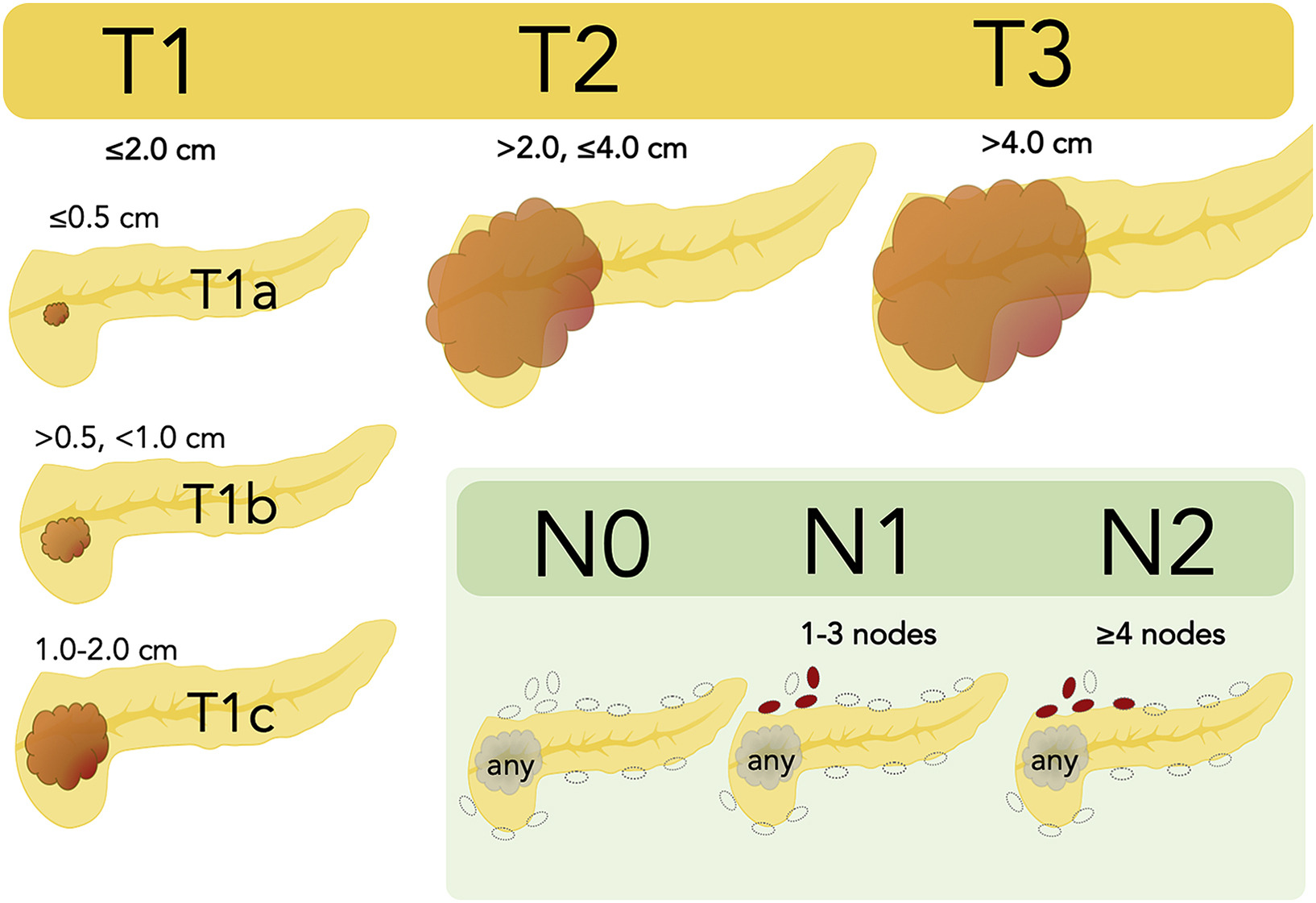
TNM Classification of Pancreatic Cancer
Treatment Approaches
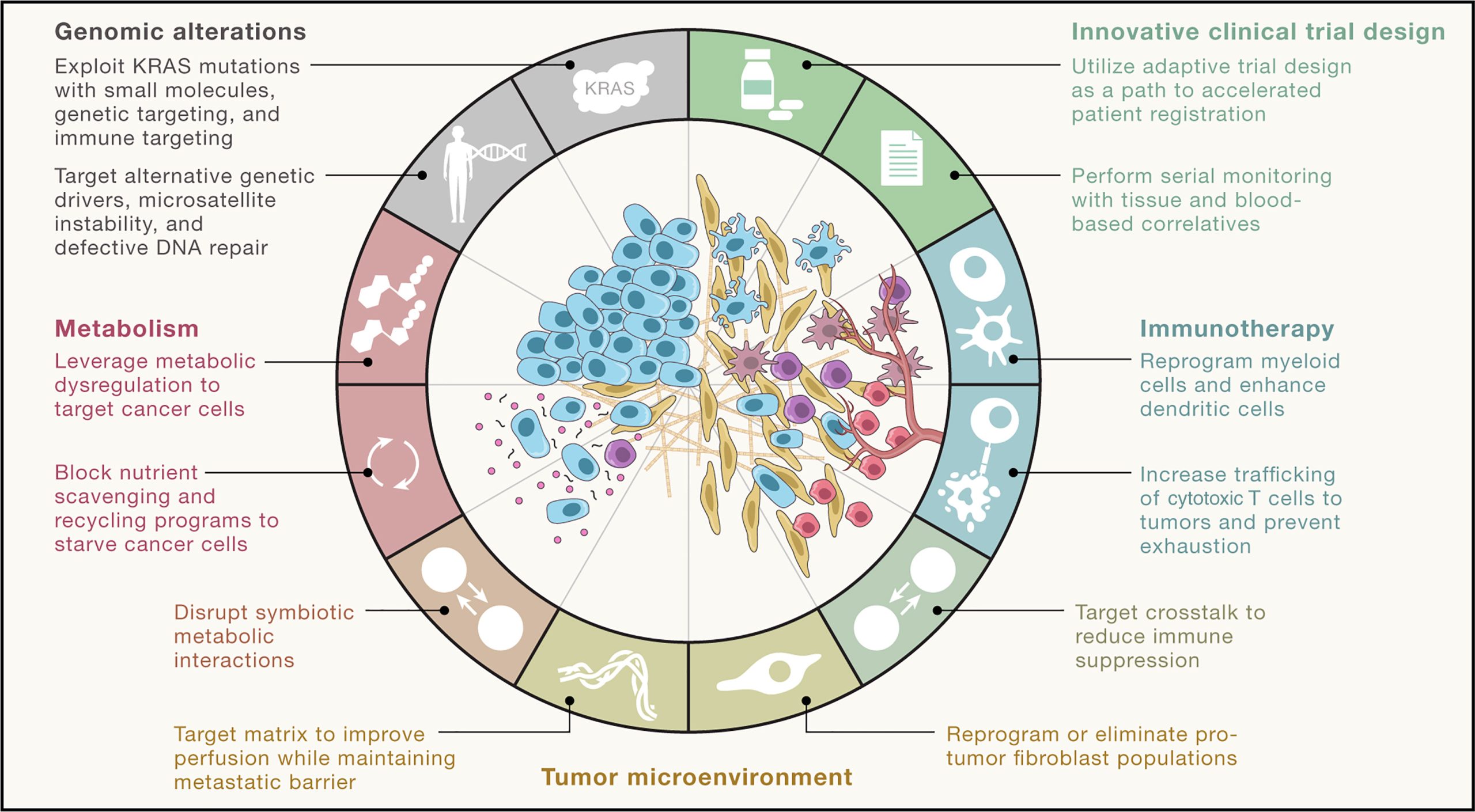
Management depends on the stage:
- Resectable disease:
- Surgical removal via the Whipple procedure
- Followed by adjuvant chemotherapy
- Locally advanced disease:
- Chemotherapy with or without radiotherapy
- Metastatic disease:
- Systemic chemotherapy (e.g., FOLFIRINOX)
Prognosis
PDAC has a 5-year survival rate of less than 10%, making it one of the deadliest cancers. The main challenges include:
- Late presentation
- Early metastasis
- Limited effectiveness of traditional therapies
RAS AND PANCREATIC CANCER
Under normal conditions, RAS is activated by signals from cell surface receptors such as receptor tyrosine kinases (RTKs). Once activated, it triggers downstream pathways, most notably:
- The MAPK (RAF–MEK–ERK) pathway, which promotes cell proliferation
- The PI3K–AKT pathway, which enhances cell survival and metabolism
This tightly controlled system ensures proper cellular function and tissue homeostasis.
When mutated, RAS remains locked in its active (GTP-bound) form, continuously sending growth and survival signals. This leads to uncontrolled cell division, resistance to apoptosis, and ultimately tumor development.
New classes of drugs are being developed, that target RAS proteins, like :
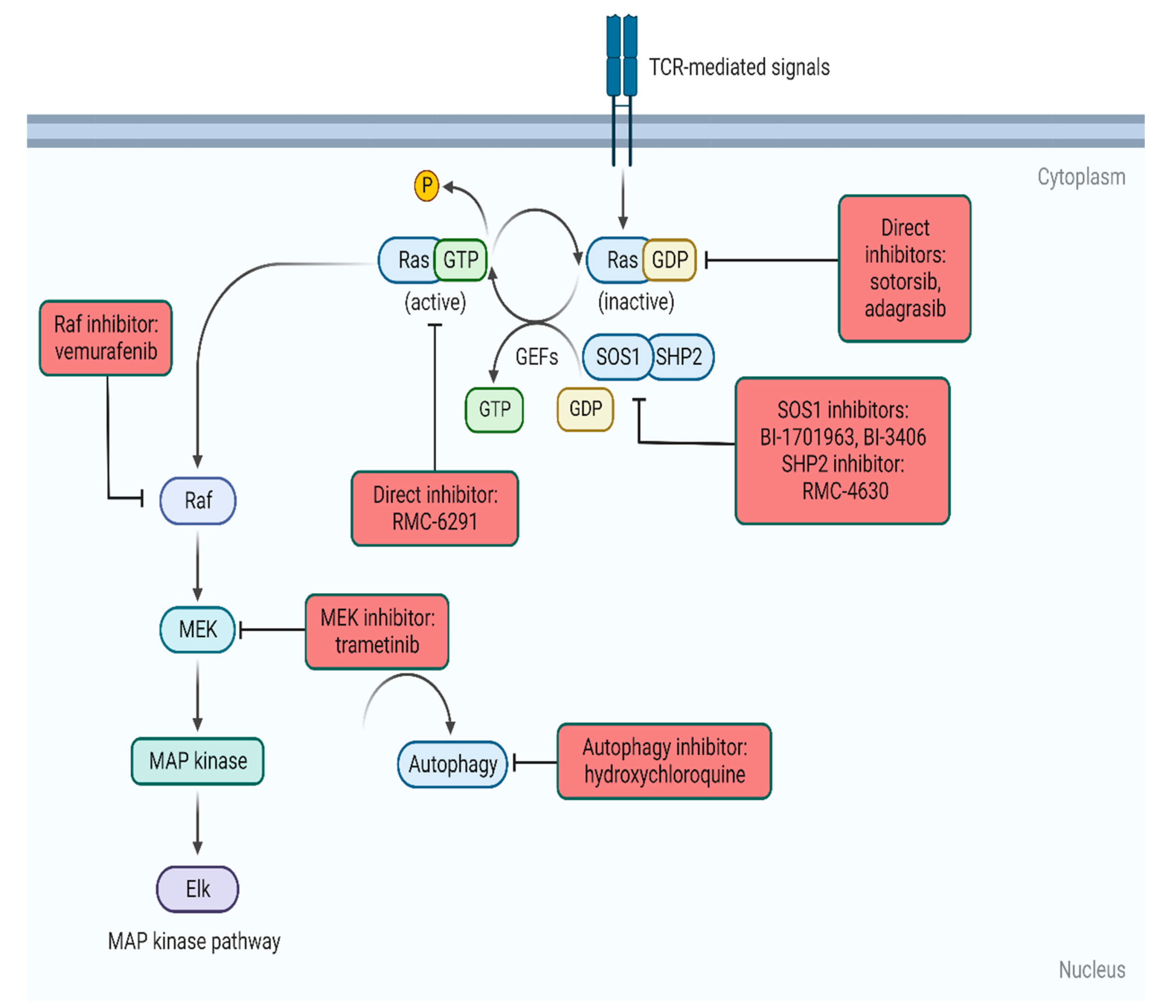
Different Drug Action Sites
A NOVEL BREAKTHROUGH
An experimental drug called daraxonrasib (RMC-6236) developed by the biotechnology company Revolution Medicine shows remarkable result in the clinical trials. This drug targets the RAS pathway, a key driver of cancer growth present in over 90% of pancreatic tumors as discussed above.
In a recent Phase 3 clinical trial, daraxonrasib demonstrated remarkable outcomes in patients with advanced pancreatic cancer:
- Median overall survival:
- 13.2 months with the drug
- 6.7 months with standard chemotherapy
- Significant improvement in progression-free survival
- Effective across multiple RAS mutation type
This represents nearly a doubling of survival, a rare achievement in pancreatic cancer treatment.
Riding on the current momentum, multiple trials are being performed :
- RASolute 302 → metastatic PDAC (positive Phase 3 results)
- RASolute 303 → first-line treatment setting
- RASolute 304 → post-surgery (adjuvant therapy) aiming to improve disease-free survival
Official Title
RASolute 304: A Phase 3 Multicenter, Open-label, Randomized, 2-Arm Study of Adjuvant Daraxonrasib Versus Standard of Care Observation Following Completion of Neoadjuvant and/or Adjuvant Chemotherapy in Patients With Resected Pancreatic Ductal Adenocarcinoma (PDAC)
Study Overview
This is a global, randomized, open-label, Phase 3 study designed to evaluate whether treatment with daraxonrasib will improve disease-free survival (DFS) compared to SOC observation in patients with resected PDAC who have completed neoadjuvant and/or adjuvant chemotherapy.
Participation Criteria
Inclusion criteria is :
- At least 18 years old and has provided informed consent.
- Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1.
- Histologically confirmed PDAC with successful (R0/R1) curative intent surgical resection and no evidence of recurrent or metastatic disease.
- Must have received perioperative (neoadjuvant, adjuvant, or a combination of both) multi-agent chemotherapy.
- Must have completed most recent treatment within the past 12 weeks.
- Adequate organ function (bone marrow, liver, kidney, coagulation).
- Documented RAS mutation status.
- Able to take oral medications.
Criteria for Exclusion are :
- Prior therapy with direct RAS-targeted therapy (eg. degraders and/or inhibitors).
- Any conditions that may affect the ability to take or absorb study drug.
- Major surgery within 28 days prior to randomization.
- Patient is unable or unwilling to comply with protocol-required study visits or procedures.
Adults 18 years and older of all sexes are accepted. Healthy volunteers are not accepted.
Study Plan
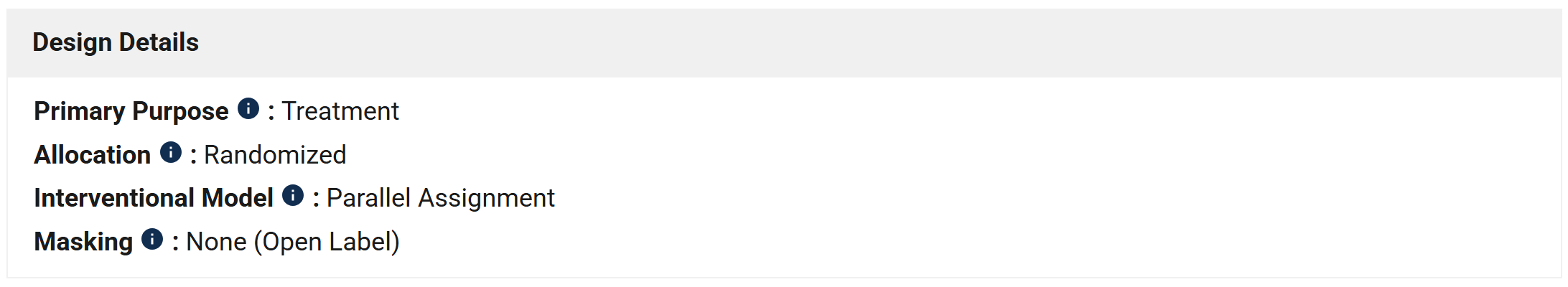

Arms and Intervention
Outcome Measures

Primary Outcome Measures
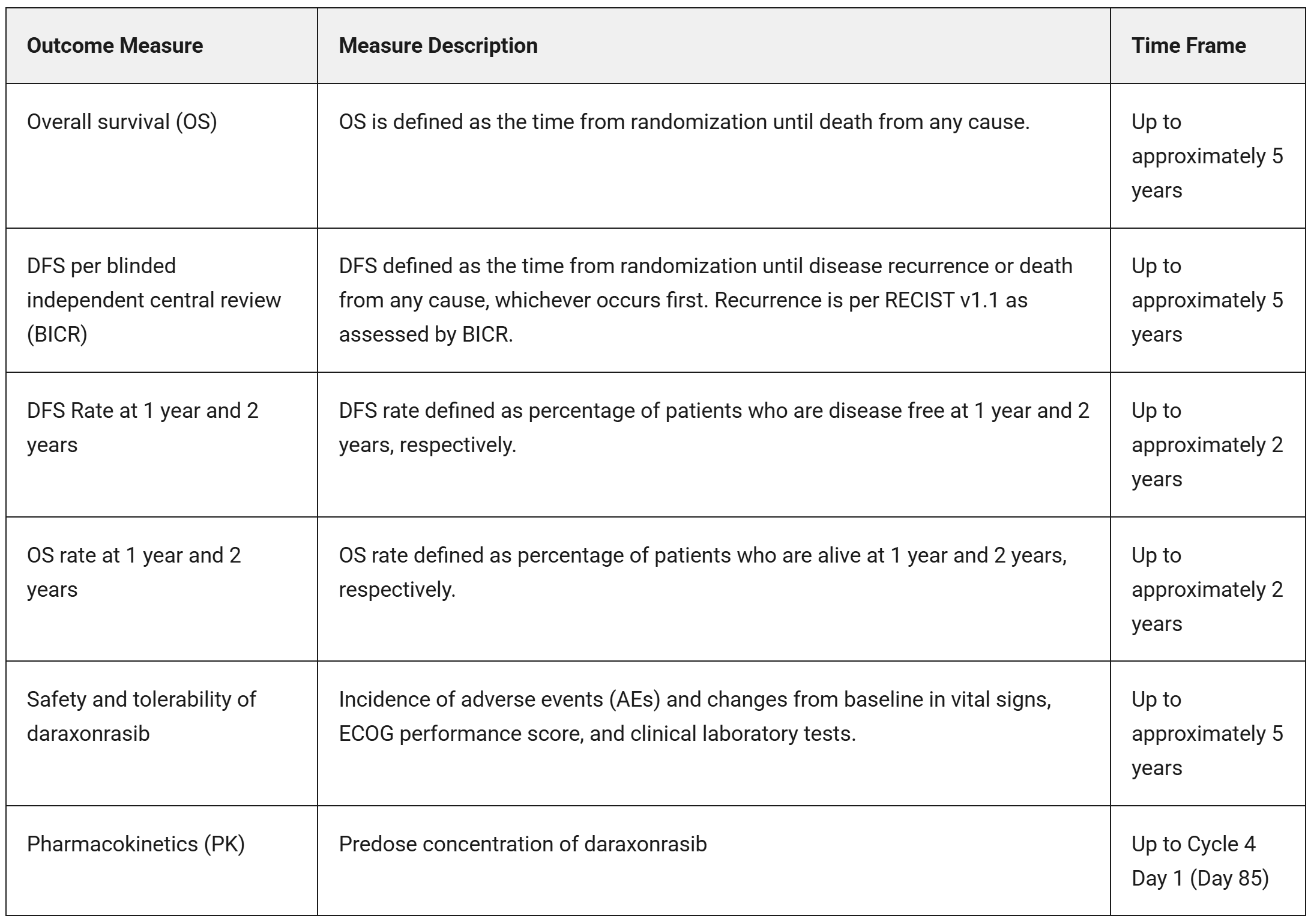
Secondary Outcome Measures
REFERENCES
https://doi.org/10.1016/j.ejso.2020.02.014.

0 comments